lv lf physics | lf vs lv lv lf physics Lf and Lv are collectively called latent heat coefficients. They are latent, or hidden, because in phase changes, energy enters or leaves a system without causing a temperature change in the system; so, in effect, the energy is hidden. Table 1 . Watch Straps Compatible with Omega Silicone 20mm 22mm Soft Replacement Straps Bracelet for Watches for Omega Seamaster 300 SpeedMaster De Ville OMEGA Ocean Universe 3.5 out of 5 stars 27 £25.54 £ 25 . 54
0 · lf vs lv
1 · lf and lv phase change
2 · lf and lv fusion
A*Men Pure Malt - Création 2013 Mugler 2013. See Prices. 8.2 / 10 153 Ratings. A popular limited perfume by Mugler for men, released in 2013. The scent is sweet-gourmand. Projection and longevity are above-average. It was last marketed by Clarins. Limited Edition. More.A*Men Pure Malt Creation by Thierry Mugler is a limited edition fusion between Amen and the world of sprits thanks to a patented method inspired by traditional techniques used to .
Latent Heat of Fusion has the symbol Lf. The units of Latent Heat of Fusion are J/kg (Joules per kilogram) Lf is the heat energy required to change. 1kg of a solid (at melting point) into 1kg of .Going through the process of making your own ice cream lets you invent your own flavors and marvel at the physics firsthand (Figure 11.11). The first step to making homemade ice cream is .
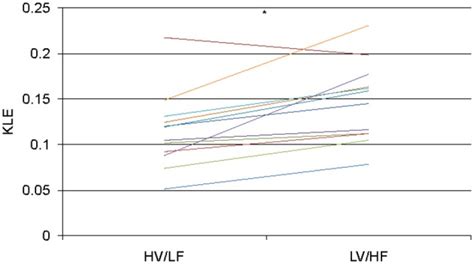
The heat Q required to change the phase of a sample of mass m is given by Q = mLf Q = m L f (melting or freezing) and Q = mLv Q = m L v (evaporating or condensing), where Lf L f and Lv .Lf and Lv are collectively called latent heat coefficients. They are latent, or hidden, because in phase changes, energy enters or leaves a system without causing a temperature change in the system; so, in effect, the energy is hidden. Table 1 .Latent Heat of Fusion has the symbol Lf. The units of Latent Heat of Fusion are J/kg (Joules per kilogram) Lf is the heat energy required to change. 1kg of a solid (at melting point) into 1kg of .Latent heat of a substance is the amount of energy absorbed or released by the substance during a change in its physical state that occurs without changing its temperature. SI unit of latent heat is the joule ().
lf vs lv
Going through the process of making your own ice cream lets you invent your own flavors and marvel at the physics firsthand (Figure 11.11). The first step to making homemade ice cream is to mix heavy cream, whole milk, sugar, and your flavor of choice; it could be as simple as cocoa powder or vanilla extract, or as fancy as pomegranates or .Lf and Lv are collectively called latent heat coefficients. They are latent, or hidden, because in phase changes, energy enters or leaves a system without causing a temperature change in the system; so, in effect, the energy is hidden. Table 1 lists representative values of Lf and Lv, together with melting and boiling points.The heat Q required to change the phase of a sample of mass m is given by Q = mLf Q = m L f (melting or freezing) and Q = mLv Q = m L v (evaporating or condensing), where Lf L f and Lv L v are the latent heat of fusion and the latent heat of vaporization, respectively.
(evaporating or condensing), where Lf and Lv are the latent heat of fusion and the latent heat of vaporization, respectively. Key Terms. latent heat of fusion: the energy required to transition one unit of a substance from solid to liquid; equivalently, the energy liberated when one unit of a substance transitions from liquid to solid.The specific latent heat (L) of a material.. is a measure of the heat energy (Q) per mass (m) released or absorbed during a phase change. is defined through the formula Q = mL. is often just called the "latent heat" of the material. uses the SI unit joule per kilogram [J/kg].For a transition from solid to liquid, L is known as the latent heat of fusion, Lf, and for a liquid to gas transition, it is known as the latent heat of vaporization, Lv. For water, the latent heat of fusion is Lf = 79.6 calories/gram. 3.3 Procedure.The latent heat of fusion (associated with the melting of a solid) is defined by: Lf = Heat required to melt 1g of solid. Thus, to melt a mass m of ice it takes an amount of energy Q = mLf. The task of this laboratory will be to determine Lf for water using calorimetry.
Introduction to Dynamics: Newton’s Laws of Motion; 4.1 Development of Force Concept; 4.2 Newton’s First Law of Motion: Inertia; 4.3 Newton’s Second Law of Motion: Concept of a System; 4.4 Newton’s Third Law of Motion: Symmetry in Forces; 4.5 Normal, Tension, and Other Examples of Forces; 4.6 Problem-Solving Strategies; 4.7 Further Applications of Newton’s .Latent Heat of Fusion has the symbol Lf. The units of Latent Heat of Fusion are J/kg (Joules per kilogram) Lf is the heat energy required to change. 1kg of a solid (at melting point) into 1kg of .Latent heat of a substance is the amount of energy absorbed or released by the substance during a change in its physical state that occurs without changing its temperature. SI unit of latent heat is the joule ().
Going through the process of making your own ice cream lets you invent your own flavors and marvel at the physics firsthand (Figure 11.11). The first step to making homemade ice cream is to mix heavy cream, whole milk, sugar, and your flavor of choice; it could be as simple as cocoa powder or vanilla extract, or as fancy as pomegranates or .Lf and Lv are collectively called latent heat coefficients. They are latent, or hidden, because in phase changes, energy enters or leaves a system without causing a temperature change in the system; so, in effect, the energy is hidden. Table 1 lists representative values of Lf and Lv, together with melting and boiling points.
The heat Q required to change the phase of a sample of mass m is given by Q = mLf Q = m L f (melting or freezing) and Q = mLv Q = m L v (evaporating or condensing), where Lf L f and Lv L v are the latent heat of fusion and the latent heat of vaporization, respectively.
(evaporating or condensing), where Lf and Lv are the latent heat of fusion and the latent heat of vaporization, respectively. Key Terms. latent heat of fusion: the energy required to transition one unit of a substance from solid to liquid; equivalently, the energy liberated when one unit of a substance transitions from liquid to solid.The specific latent heat (L) of a material.. is a measure of the heat energy (Q) per mass (m) released or absorbed during a phase change. is defined through the formula Q = mL. is often just called the "latent heat" of the material. uses the SI unit joule per kilogram [J/kg].For a transition from solid to liquid, L is known as the latent heat of fusion, Lf, and for a liquid to gas transition, it is known as the latent heat of vaporization, Lv. For water, the latent heat of fusion is Lf = 79.6 calories/gram. 3.3 Procedure.
The latent heat of fusion (associated with the melting of a solid) is defined by: Lf = Heat required to melt 1g of solid. Thus, to melt a mass m of ice it takes an amount of energy Q = mLf. The task of this laboratory will be to determine Lf for water using calorimetry.
lf and lv phase change
lf and lv fusion
louis vuitton die press tdp
Oversized Women's Active Zip up Hoodie Lightweight Comfortable Long Sleeve .
lv lf physics|lf vs lv